Reprocessed Single Use Device

The EU Medical Device Regulation (MDR) introduced stricter rules and clearer responsibilities for all actors involved in the life cycle of medical devices. One area that raises particular questions is…

The EU Medical Device Regulation (MDR) introduced stricter rules and clearer responsibilities for all actors involved in the life cycle of medical devices. One area that raises particular questions is…
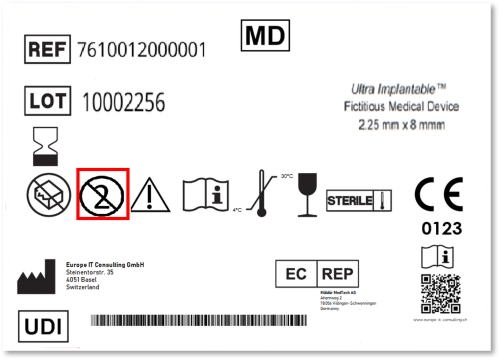
Since the EU Medical Device Regulation (MDR) came into force, the Unique Device Identification (UDI) system has become a cornerstone of regulatory compliance. For manufacturers of Single Use Devices (SUDs),…