Since the EU Medical Device Regulation (MDR) came into force, the Unique Device Identification (UDI) system has become a cornerstone of regulatory compliance. For manufacturers of Single Use Devices (SUDs), there are specific requirements that must be carefully understood and implemented—especially when it comes to EUDAMED registration.
What is a Single Use Device?
A Single Use Device is a medical device intended by the manufacturer to be used only once for a single patient during a single procedure, and not to be reused or reprocessed.
Common examples include:
- Disposable syringes
- Catheters
- Surgical masks
- Blood lancets
Such products require special attention in both labelling and UDI data handling.
UDI Obligations for Single Use Devices
Like all medical devices under the MDR, single-use products must comply with the UDI requirements. However, there are unique aspects:
-
Labeling and Packaging:
Most single-use products are either too small for direct labeling or sold in bulk. In those cases, the UDI-DI and UDI-PI are typically placed on the outer packaging. -
EUDAMED Registration:
Single Use Devices must be registered in EUDAMED, including the Basic UDI-DI and device-specific data, just like reusable devices.
“Labelled as Single Use Device” – What Does It Mean?
In EUDAMED, it must be explicitly declared whether the product is labelled as single-use. This is done using the field:
“Labelled as a Single Use Device” = TRUE
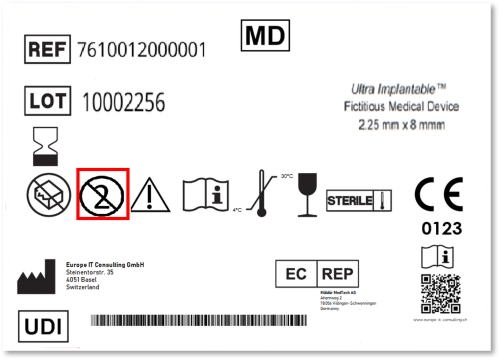
This means the product label must clearly state that it is intended for single use only, either through standard symbols (e.g., the crossed-out “2” symbol indicating “Do not reuse”) or written text (“Single use only” / “Do not re-use”).
⚠️ Important: This is not just about how the product is used but how it is labelled and declared under MDR and the UDI Core Data Set requirements.
Impact on Other UDI Attributes
When a device is labelled as single-use, several other EUDAMED fields must align accordingly:
| EUDAMED Field | Typical Value for SUDs |
|---|---|
| Labelled as Single Use Device | TRUE |
| Reusable | FALSE |
| Has Serial Number | FALSE (in most cases) |
| Device is Reprocessed | FALSE |
| Sterile | TRUE or FALSE depending on product |
This ensures consistency across the UDI data set and helps avoid registration errors or delays.
Why It Matters for Manufacturers
Incorrectly labelling or misdeclaring a device’s single-use status can:
- Lead to EUDAMED validation issues
- Create regulatory risks during audits
- Affect traceability and market access
Clear, compliant labelling and accurate data entry are essential to avoid these pitfalls.
Our Recommendation
Manufacturers should ensure:
- Product labels explicitly indicate single-use status (text or symbol),
- UDI attributes in EUDAMED are fully aligned,
- Packaging hierarchy and labeling strategy are optimized for UDI compliance.






Related Posts