Medical Devices Market in UK

Opportunities, Challenges, and Regulations The United Kingdom (UK) remains a significant market for medical devices despite its exit from the European Union (Brexit). With a growing healthcare sector, investments in…

Opportunities, Challenges, and Regulations The United Kingdom (UK) remains a significant market for medical devices despite its exit from the European Union (Brexit). With a growing healthcare sector, investments in…

Medical Mountains, the leading network for medical technology and life sciences in Tuttlingen, warmly invites you to the seminar “Implementation of the EU-MDR – EUDAMED”! This expert seminar offers in-depth…
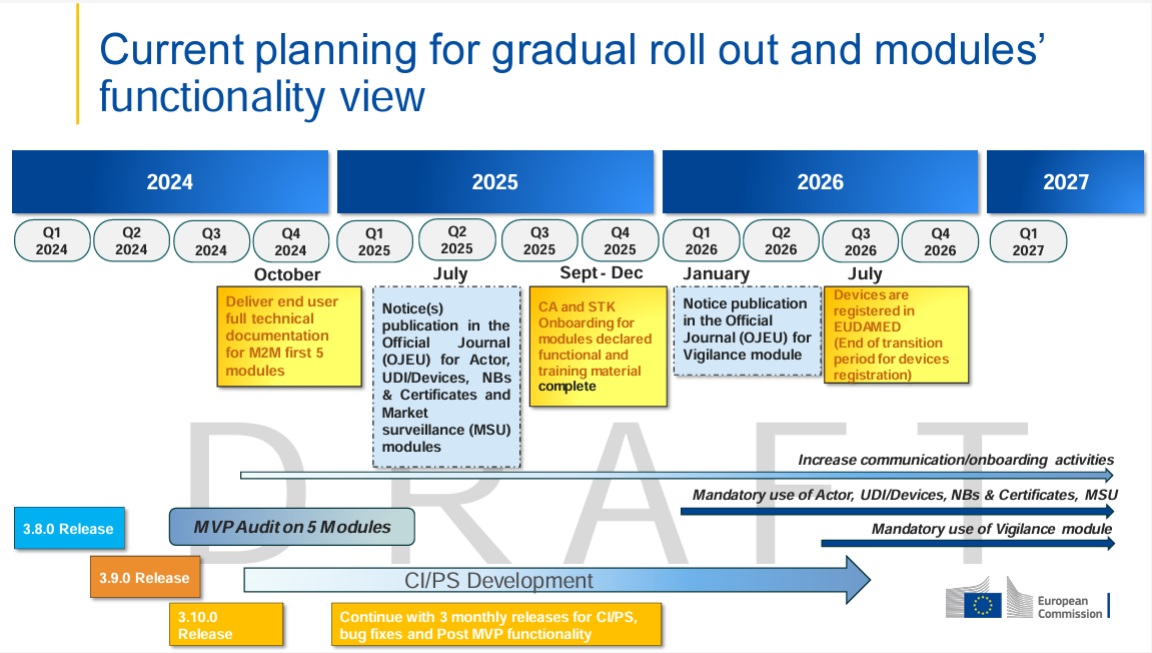
We would like to inform you today about the latest developments regarding the Vigilance module in the EUDAMED database. This crucial module, scheduled to go live in 2026, will be…

The FDA recently announced the launch of its Next Generation Electronic Submissions Gateway (ESG) interface, aiming to enhance the efficiency and user experience of electronic submissions for regulatory documents, including…

Time is running out for the medical device industry to comply with the requirements of the Medical Device Regulation (MDR). To properly implement such complex regulations, it's essential to have…

Opportunities, Challenges, and Regulations India is one of the fastest-growing markets for medical devices globally. With an annual growth rate of approximately 16-17%, it presents significant opportunities for both domestic…

Successful Audit Completion at EUROPE IT Consulting GmbH Dear Business Partners, Colleagues, and Customers, We are pleased to announce that EUROPE IT Consulting GmbH has successfully passed the recent audit.…

Bern, Switzerland - August 6, 2024 Swissmedic has announced the successful go-live of the Swissdamed Actors Module today. This marks a significant milestone in the regulatory framework for medical devices…
Basel, Switzerland – We at Europe IT Consulting GmbH are thrilled to announce a significant milestone in our ongoing efforts to support medical device manufacturers with compliance and regulatory requirements. (more…)

Modern Outsourcing Strategies for Your Business Welcome to EUROPE IT Consulting, your reliable partner for efficient and cost-effective IT solutions. In today's globalized world, there are various approaches to outsourcing…