EUDAMED Database Update App-Version 2.7.0

On April 4th, the EUDAMED was updated. With this release, some known errors have been corrected, which were reported by Europe IT Consulting GmbH, among others. But the main innovation…

On April 4th, the EUDAMED was updated. With this release, some known errors have been corrected, which were reported by Europe IT Consulting GmbH, among others. But the main innovation…
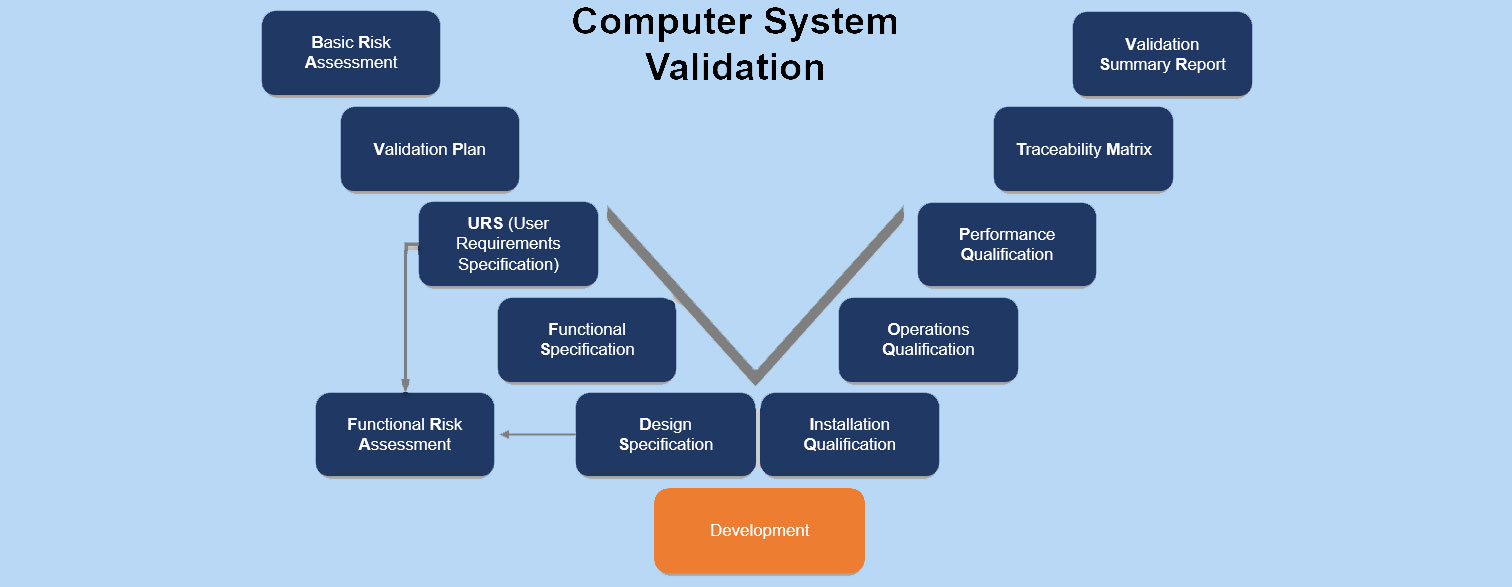
Computer System Validation - CSV Computer System Validation refers to a documented process that ensures a computerized system does exactly what it was designed to do, in a consistent and…

Medical device actors can gives their views and contribute to the future UK´s regulatory framework. Since September 16, 2021, the Medical and Healthcare products Regulatory Agency (MHRA) gives the opportunity…

Submissions for PECP (Performance Evaluation Consultation Procedure) are accepted since September 3, 2021, by the expert panels in the field of in vitro diagnostic medical devices. What are the expert…

New differing requirements now apply to EU and Swiss medical device manufacturers.As already mentioned in a previous article (MRA between Switzerland and EU no longer valid), Switzerland is considered as…
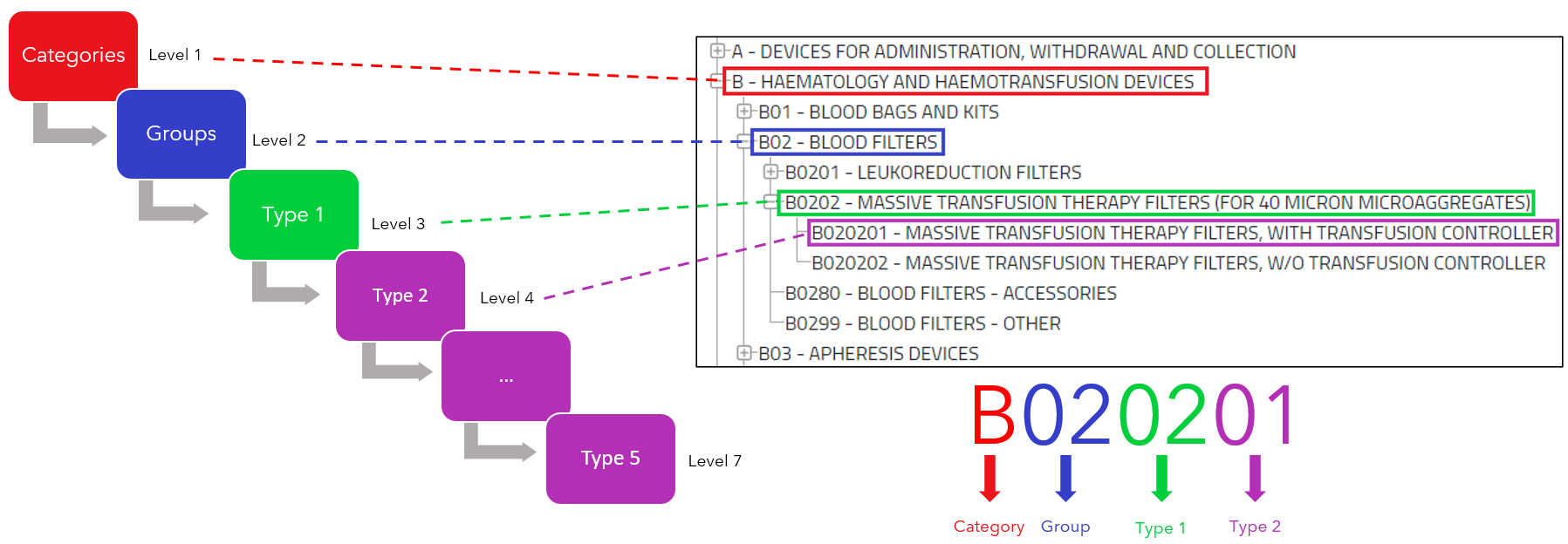
With the implementation of the Medical Device Regulation comes the new EMDN (European Medical Device Nomenclature), as stated in the regulations (Art.26 2017/745 MDR, Art.23 2017/746 IVDR). Review this concept…
UDI EUDAMED Overview With the new Medical Device Regulation (MDR 2017/745) and In-Vitro Diagnostic Device Regulation (IVDR 2017/746) of the European Commission, a new requirements for medical devices were introduced.…
Dear Sir or Madam,You have participated in our UDI EUDAMED Webinar 2021, so you are aware of the challenges medical device manufacturers face with the Medical Device Regulation of MDR2017/745…

The Medical Device Regulation (MDR) is about to apply in the European Union, starting on 26 May 2021. Since Switzerland is not a member state, this regulation does not apply…
UDI EUDAMED Training The implementation of the European Commission's Medical Device Regulation (MDR 2017/745 & IVDR 2017/746) is a major challenge for all medical device manufacturers, importers and distributors. Due to…